
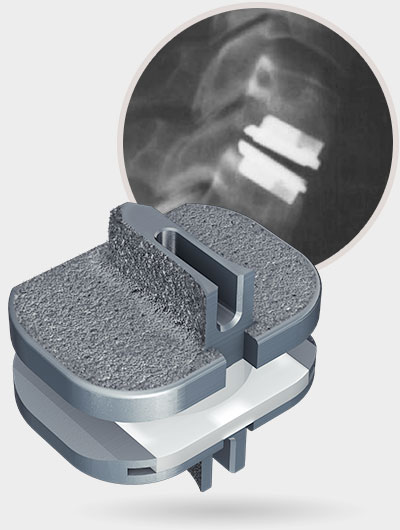

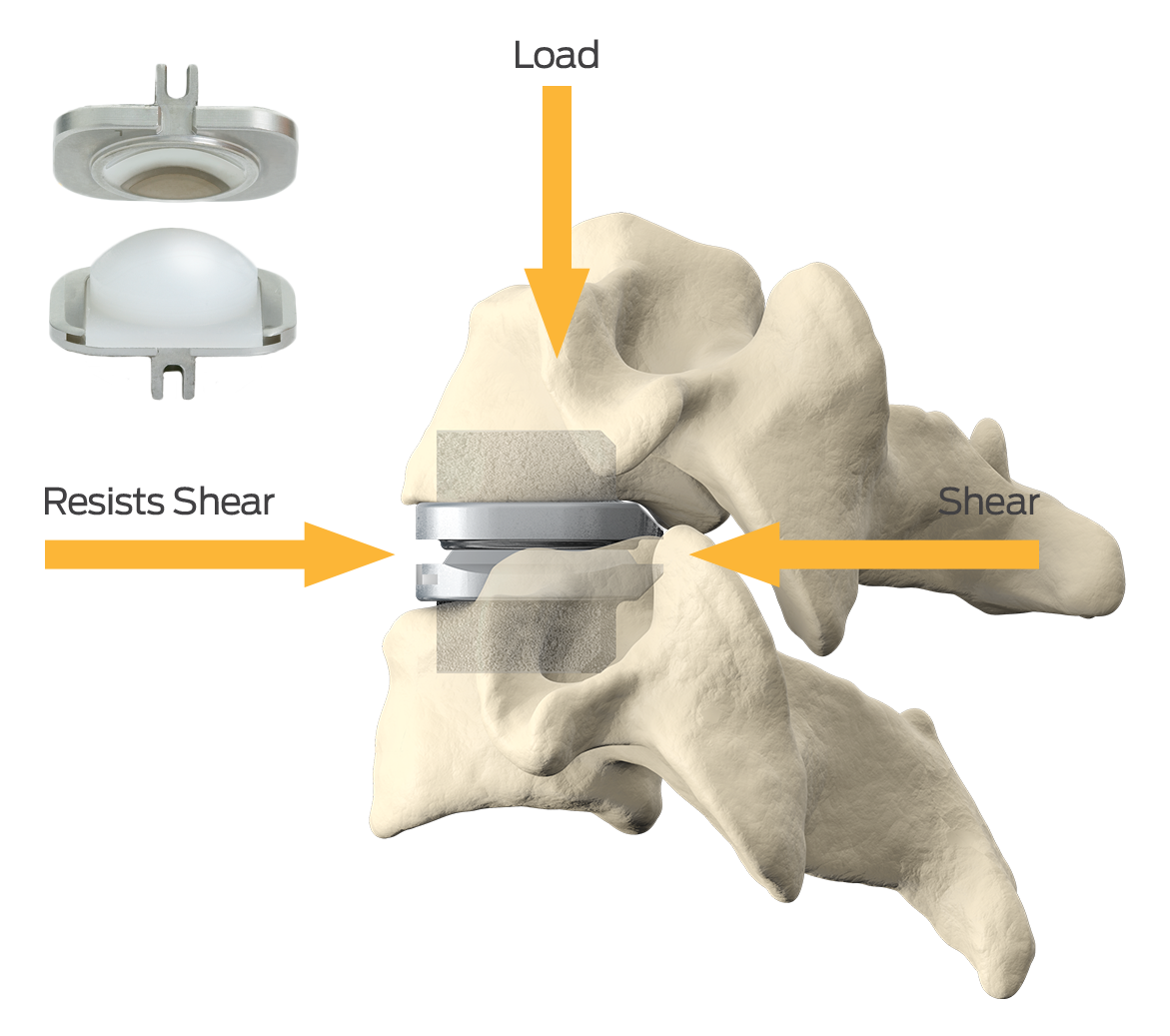

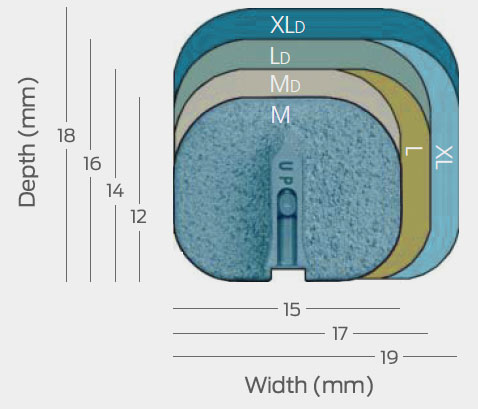
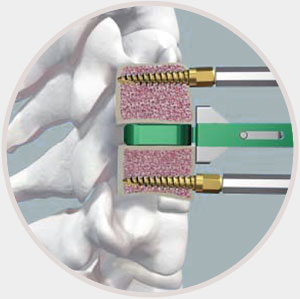


1 Data on file for all prodisc cervical and lumbar devices.
2 Search performed on Pubmed, Embase, Ovid Medline® covering 1988 – 2025.
3 Janssen ME, et al, ProDisc-C Total Disc Replacement Versus ACDF for Single-Level Symptomatic Cervical Disc Disease, JBJS, 2015, 97:1738-47.
4 Zigler J, et al, Five-year adjacent-level degenerative changes in patients with single-level disease treated using lumbar total disc replacement with ProDisc-L versus circumferential fusion, J Neurosurg Spine 17(6):504–511, 2012.
Watch this short primer on Centinel Spine and its unique and extraordinary place as a catalyst of change in the spine industry—with pioneering technologies and a clinical history that have led to successes ranging from PGA champions to a growing list of surgeon-patients.
 SEE MORE VIDEOS
SEE MORE VIDEOS